  Color table with atomic numbers, element symbols, element names, atomic weights, periods, and groups. Chemical formula, (CH)SO Molar Mass, 78.13 g/mol HS Code, 2930 90 98 Quality Level, MQ100 Application, Dimethyl sulfoxide for gas chromatography. That’s why our solvents for DNA and peptide synthesis are carefully processed and purified to optimize efficiency and yields in these critical processes.Ä¡ Strongly contaminated halogen-free organic solvents: container A. Color Printable Periodic Table - Pretty much everything you need that can fit on a page and still be readable. 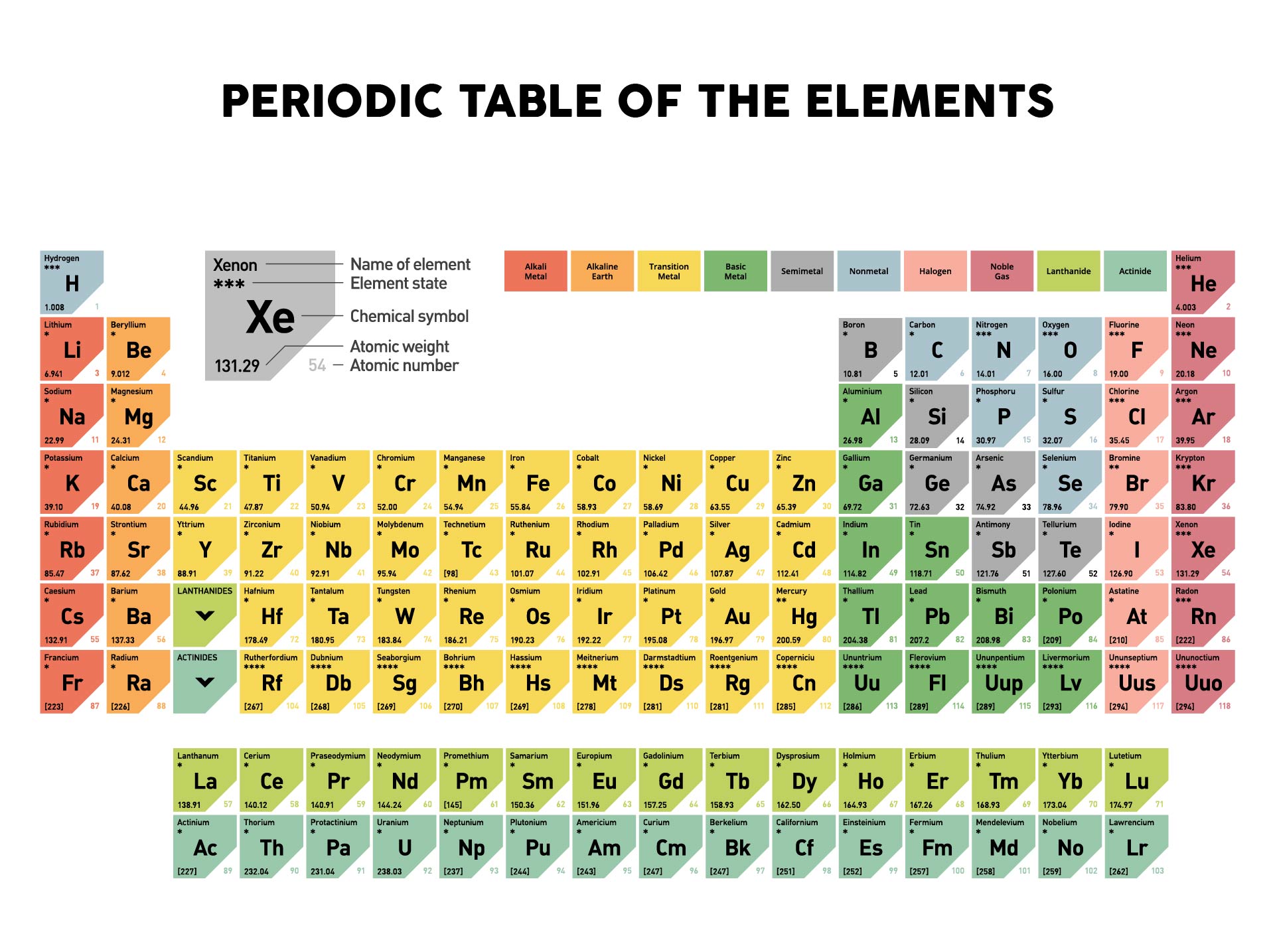 The periodic table lists the atomic mass of carbon as 12.011 amu the average molar mass of carbonthe mass of 6.022 × 10 23 carbon atomsis therefore 12. dimethyl sulfoxide DMSO 67-68-5 Methyl sulfoxide Methylsulfinylmethane. periodic refills of that amount if all of the following requirements are satisfied. CAS, molar mass 84.17 g/mol.Ä¡ Strongly contaminated halogen-free organic solvents: container A.Methanesulfinylmethane, Methyl sulfoxide, Dimethyl(oxido)sulfur, DMSOÄNA and peptide synthesis applications demand solvents with the lowest possible water content. 20Chol membrane system with 5 mol DMSO, although the general features are preserved for other solvent compositions. The molar mass of any substance is its atomic mass, molecular mass, or formula mass in grams per mole. periodic boundary conditions, and no center of mass motion removal: temperature T1. In case of all the water soluble deuterated standard products, water content is specified according to both Karl Fischer and NMR.Äimethyl sulfoxide-D6 with TMS (0.1 vol.%), deuteration degree min. Print the total mass of atoms in (in amu) for selected topology.In this method, the problem of removing the water formed during the reaction is overcome with the selected organic solvent. Depending on application and sensitivity of the NMR spectrometer our offer solvents with deuteration degrees between 98% and 99.96%. High molecular weight-PGA (M w > 930,000 g/mol) can be obtained by polycondensation in azeotropic solution called azeotropic condensation polymerization 22, 28,29,30. Click Table Columnsto select which columns to display. Add them together: add the results from step 3 to get the total molar mass of the compound. Calculate molar mass of each element: multiply the atomic mass of each element by the number of atoms of that element in the compound. Hexadeuterodimethyl sulfoxide, DMSO deuteratedĪ wide range of MagniSolv â„¢ deuterated solvents with extremely low residual water, excellent chemical purity, and the highest isotopic enrichment available can satisfy the most demanding requirements of researchers. The report point table does not list response values adjusted for molecular weight or capture level. American Chemical Society 57-71 Lithium 6.94 Beryllium 9.012 Sodium 22.99 Magnesium 24.31 Francium (223) Radium (226) Rutherfordium (265) Dubnium. The atomic mass is usually found on the periodic table and is given in atomic mass units (amu). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
